The complex shape of respiratory syncytial virus is one hurdle limiting the development of treatments for an infection that leads to hospitalization or worse for hundreds of thousands of people in the United States each year, according to the Centers for Disease Control and Prevention. New images of the virus from researchers at the University of Wisconsin–Madison may hold the key to preventing or slowing RSV infections.
RSV is of greatest concern in young children, the elderly and adults at high risk for respiratory complications. Yet unlike the flu and other common, communicable respiratory illnesses that annually sweep through schools, there are few options for fighting RSV. In the U.S., prophylactic treatments are available for young children, and existing vaccines are approved only for pregnant women and the elderly.
READ MORE: Scientists pinpoint new drug target for RSV
READ MORE: New RSV vaccines on the way - so what do we do about vaccine hesitancy?
The virus’s structure — which consists of tiny, bending filaments — have eluded researchers. This has made it difficult to identify key drug targets, including viral components that are conserved across related viruses.
Related viruses
“There are a number of viruses related to RSV that are also significant human pathogens, including measles,” says Elizabeth Wright, a UW–Madison biochemistry professor. “What we know about related viruses gives us clues about RSV protein structures, but to identify drug targets we need a closer look at RSV proteins that are intimately associated with the membranes of host cells.”
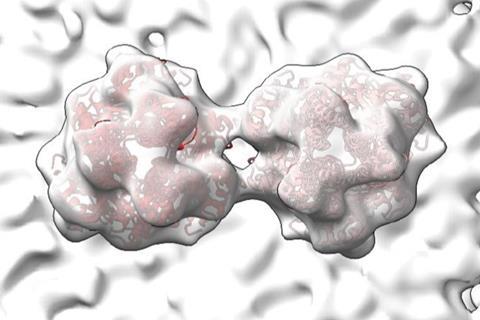
Using an imaging technique called cryo-electron tomography, Wright and her team have now revealed details of molecules and structures essential to RSV’s form and function. They published their findings recently in Nature.
Cryo-ET freezes viral particles or other molecules at ultracold temperatures, stopping biological processes in action. This allows researchers to examine the structures of organisms, cells and organelles, and viruses and capture small-scale images of structures frozen in time. Flash-freeze many RSV particles, and cryo-ET imaging will capture (nearly) all the virus’s possible configurations from many different angles. These 2D images are combined to produce a representation of the virus’s 3D structures at high resolutions — even at the level of individual atoms.
Two crucial proteins
Wright’s recent study produced high-resolution images detailing the structure of two RSV proteins, RSV M protein and RSV F protein, that are crucial to the interaction between the virus and the host cell membrane. Both proteins are also present in related viruses.
RSV M protein interacts with host cell membranes, holding together the virus’s filamentous structure and coordinating viral components and other proteins — including RSV F proteins. RSV F proteins sit on the viral surface, ready to engage with host cell receptors and regulate the virus’s fusion and entry into the host cell. The scientists’ images reveal that in RSV, two F proteins come together to form a more stable unit. Wright says that this association may prevent the F proteins from prematurely infecting the host cell.
“Our primary findings reveal structural details that allow us to better understand not only how the protein regulates assembly of viral particles, but also the coordination of proteins that enable the virus to be infectious,” says Wright.
The scientists believe that F protein pairs may be a key to destabilizing the virus before it is ready to infect its next host, making pairs of F proteins a possible target for future drug development. They will continue to explore how RSV proteins interact with each other to cause infection.






No comments yet