Researchers at Umeå University and Tartu University have found that a history of repeated antibiotic use causes defects in the normally protective mucus barrier of the gut, due to antibiotic-driven alterations in the microbiota. In a further study, the researchers found a bacteria-independent mechanism through which antibiotics can damage the mucus barrier directly.
The results have been published in the scientific journals Gut Microbes and Science Advances.
“Together, these two studies suggest that antibiotics can damage the mucus layer through at least two independent mechanisms, and that they may have long-lasting effects through an altered gut bacteria. This further supports the notion that antibiotics should be administered in a responsible manner,” says Björn Schröder, Docent in Infection Biology in the Department of Molecular Biology at Umeå University.
Risk to health
While antibiotics are undoubtedly an invaluable resource for fighting bacterial infection, it is becoming increasingly clear that they can also present a serious risk to our health if overused or misused, including causing problems in our guts. Previous research has shown the consequences of short-term antibiotic treatments on the intestinal environment, but it is less clear how repeated antibiotic use in past years can affect our guts.
READ MORE: Alterations to gut mucus may trigger ulcerative colitis
READ MORE: Study reveals possible triggers for inflammatory bowel disease
To address this question, Björn Schröder and his group at Umeå University teamed up with a research group at Tartu University in Estonia, who have built a deeply characterised cohort of individuals that provided stool samples and health records.
The researchers selected individuals who had taken at least five courses of antibiotics in the past, but not within six months before the stool collection, and compared their microbiota composition to individuals who had not taken any antibiotics within the last ten years.
“The analysis revealed changes to the gut bacteria composition, even though the antibiotics were taken a long time ago. These results indicate that repeated antibiotic use has a lasting effect on gut bacteria composition that can persist at least months after the last treatment,” says Kertu-Liis Krigul, PhD student at Tartu University.
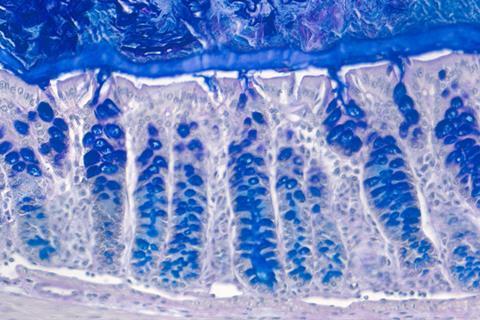
Mucus layer function
After transplantation of the human microbiota into mice and using specialised methods to analyse the mucus function in the gut, the researchers found that the function of the mucus layer was disrupted in mice transplanted with bacteria from humans with a history of repeated antibiotic use. Expansion of the mucus was reduced, and the mucus layer became penetrable, allowing bacteria to move closer to the intestinal lining.
“Looking at the bacteria present in the gut in more detail, we could see that bacteria known to feed on the mucus layer were present at higher levels in these mice. This further supports a role for the gut bacteria in determining how well the mucus barrier can function,” says Rachel Feeney, PhD student at the Department of Molecular Biology at Umeå University.
Gut bacteria
A separate study carried out in another international collaboration further showed that antibiotics can also directly disrupt the mucus barrier in a gut bacteria-independent manner.
By giving the antibiotic vancomycin to normal and ‘bacteria-free’ mice, the researchers were able to show that this antibiotic can act directly on the mucus barrier, independent of the gut bacteria. Complementary experiments on intestinal tissue were carried out at Umeå University and showed that the antibiotic could disrupt the mucus expansion within a few minutes of application.





No comments yet