The members of Bathyarchaeia are among the most abundant microorganisms on Earth and have long been considered key players in global carbon cycling. The lack of bathyarchaeial isolates until now has largely hampered direct understanding of the mechanisms of their carbon metabolism.

Scientists have now presented a successful continuous enrichment of Bathyarchaeia along with the discovery of a novel and specific methyltransferase for O-demethylation from lignin-derived aromatics. The key gene encoding this enzymatic system is widely present in genomes of Bathyarchaeia and in situ coastal sediments, which indicates an important yet unique role of Bathyarchaeia in lignin degradation.
These findings shed light on the metabolism and evolution of lignin-degrading microorganisms and provide insights into understanding microbially mediated carbon cycling in Earth environments.
Investigating Bathyarchaeia
This study was led by Prof. Fengping Wang (School of Oceanography, Shanghai Jiao Tong University). In 2015, The researchers enriched Bathyarchaeia from coastal sediment samples by adding lignin as the sole organic carbon source, followed by one year of incubation. The enrichment culture was transferred to artificial seawater medium supplemented with 5g/lkraft lignin and 50 mM sodium bicarbonate; then it was incubated at 35℃.
The growth of Bathyarchaeia was confirmed by quantitative PCR(Q-PCR) after two months of incubation. Subsequently, the growth of Bathyarchaeia was maintained in their lab for more than five years by consecutive 1:10 transfers at 2–3 months intervals.
By December 2021, one archaeon of Bathyarchaeia named Candidatus Baizosediminiarchaeumligniniphilus DL1YTT001 (Ca. B.ligniniphilus) predominated the enrichment culture, accounting for ~60% of the total abundance of 16S rRNA gene sequences (Fig.1).
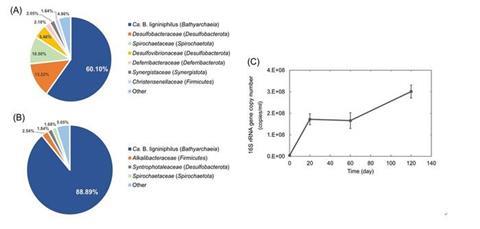
The enrichment culture was transferred to fresh media supplemented with the antibiotics as well as an additive of metabolites prepared from supernatant of the same enrichment culture. After three transfers, Ca. B. ligniniphilus reached ~90% sequence abundance of all microbes in the enrichment (Fig. 1).
Metabolome analysis
Metabolomes targeting low molecular weight (LMW) aromatic compounds on day 0 and day 30 of the incubation were analyzed by GC-MS. On day 0, a variety of lignin-derived ArOCH3, including guaiacol, vanillin, acetovanillone, vanillic acid, homovanillic acid, and syringic acid, were detected. These monomers all dramatically decreased during the 30-day incubation, and there was a substantial accumulation of their demethylated products, including catechol and protocatechoic acid; this confirmed the occurrence of O-demethylation from ArOCH3 during the incubation of Ca. B. ligniniphilus.
One gene cluster that may carry out the O-demethylation/methyltransferase process on ArOCH3 was found within the Ca. B. ligniniphilus MAG. This putative methyltransferase complex includes two copies of putative methyltransferase 1 (MT1, MtgB_1 and MtgB_2), one copy of putative methyltransferase 2 (MT2, MtgA), and one copy of putative corrinoid protein (CP,MtgC) (Fig. 2).
In the methyltransferase complex, MT1 is the key enzyme responsible for substrate selectivity and determines the type of methyl substrate.Neither of putative MT1s (MtgB_1 and MtgB_2) of Ca. B. ligniniphilus had homology with known MT1, which indicated that these two MT1s (MtgB_1 and MtgB_2) are novel.To verify the function of the novel MT1s of Ca. B. ligniniphilus in O-demethylation and methyl transfer, the MtgB_2 was expressed and purified in Escherichia coli and its O-demethylation activity was confirmed (Fig. 3).
Vertical inheritance
The genes encoding MtgB were only found in genomes of Bathyarchaeia. In 297bathyarchaeial MAGs, 60 MAGs were found to contain the Bathyarchaeia-specific MtgBgene or its gene cluster;intriguingly, all 60 MAGs clustered in the order Baizomonadales (Fig. 4). Thus, the evolution model of Bathyarchaeia-specific MtgBgenes may be vertical inheritance because they likely evolved from the common ancestor of order Baizomonadales.
The MtgB genes were identified in metagenomic datasets from a wide range of coastal sediments, and they were high expressed in coastal sediments from the East China Sea.These findings suggest that Bathyarchaeia capable of O-demethylation via their novel and specific methyltransferases are ubiquitous in coastal sediments.




No comments yet